Assent for Research in the Pediatric Intensive Care Unit (ASSENT-PICU)
CONTACT Katie O’Hearn if you are interested in joining this program of research.
SUMMARY OF THE ASSENT-PICU
The overall aim of the ASSENT-PICU research program is to establish evidence-based guidelines for assent for research in the pediatric critical care setting. Before we can move forward with guideline development, we first need to improve our understanding of what we currently do (assent practices and capacity for assent in the PICU), and how key stakeholders would like the assent process to be operationalized (perspectives of patients, caregivers, research coordinators, nurses, researchers and Research Ethics Board Chairs/Managers).
BACKGROUND AND RATIONALE
Children admitted to the PICU suffer from acute, life-threatening disease and injuries such as cardiovascular disease, respiratory failure, trauma, sepsis and multi-organ system dysfunction. During the first 24 to 48 hours of admission, these children often require significant life-saving interventions. As a result, many PICU research studies evaluate interventions administered during this initial period following PICU admission. This necessitates urgent and time-sensitive enrollment into research studies at a time when critically ill children are most likely to be clinically unstable. In this environment, there are significant barriers to asking children for their assent for research beyond their acute illness. For example: (i) PICU admission commonly results in high levels of anxiety and distress, which have been shown to have a negative impact on capacity and understanding; (ii) PICU patients are often prescribed medications that drastically alter mental status (e.g. sedative and anti-seizure medications); and (iii) PICU patients commonly require invasive mechanical ventilation thus limiting their ability to communicate. (iii) In addition, unlike outpatients, who may be followed by a single physician (giving the physician an understanding of the patient’s cognitive ability), PICU patients are typically admitted for acute reasons or admitted to PICU only while critically ill but followed by multiple other hospital services. As a result, PICU physicians often do not have the appropriate background information necessary to assess a patient’s capacity.
Due to these unique characteristics, it is extremely difficult to obtain assent for pediatric critical care research. This is true both shortly after admission, and at time of PICU discharge. In 2016-2017, our group performed an environmental scan of two PICUs in Canada and showed that at PICU admission, 81% of patients were deemed not appropriate to approach for assent most commonly due to age, influence of psychotropic medications and/or mechanical ventilation. Of the patients that were considered appropriate to approach for assent at PICU admission, the majority short-stay patients who likely would not have met eligibility criteria for ongoing PICU studies. At PICU discharge, 74% of patients were still considered not appropriate to approach, most commonly due to age and/or developmental delay. Only 8% of patients considered not approachable at admission become appropriate to approach for assent by PICU discharge.
Despite these challenges, assent remains an important part of the decision-making process around pediatric research participation. However, the assent process is currently not well defined in any pediatric population, including critically ill children. Defining and operationalizing assent in pediatric critical care research is particularly challenging for the reasons outlined above. We therefore propose a research program to better understand the perspectives of key stakeholders, as well as the perceived barriers to assent in the PICU. The end goal of this research program is to develop guidelines for assent in pediatric critical care research.
STUDIES WITHIN THE ASSENT-PICU RESEARCH PROGRAM
The ASSENT-PICU program aims to understand current assent practices and capacity for assent in the PICU, and perspectives of key stakeholders (Figure 1). We have summarized each of the studies included in this program below.
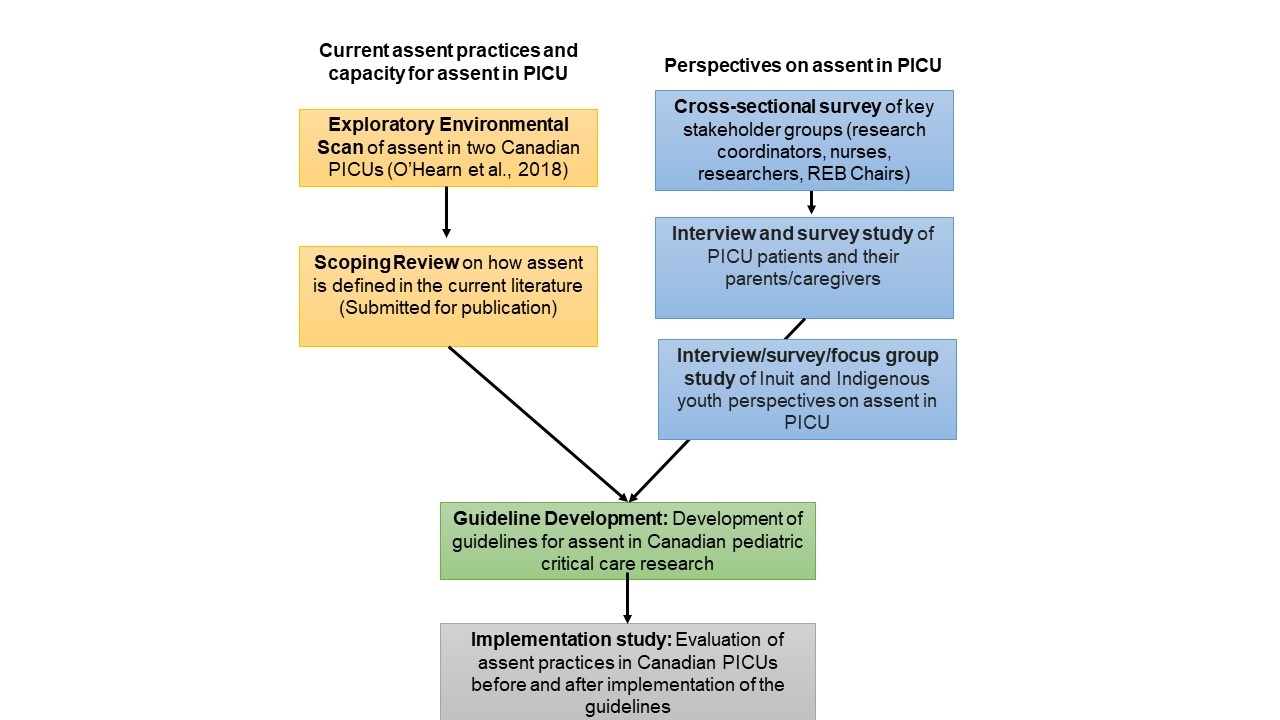
Figure 1. Overview of the ASSENT-PICU Program
ENVIRONMENTAL SCAN - Complete We conducted a pilot observational study in two PICUs to determine the proportion of PICU patients who would not be appropriate to approach for assent within the first 24 hours of admission. We also sought to determine the number of patients categorized as inappropriate to approach at admission who became appropriate to approach by discharge, the reason(s) patients were categorized as not approachable for assent, and the level of agreement of patient potential for assent at 24 hours and PICU discharge between the study team and the resident/fellow, and the study team and the bedside nurse. View the abstract for this study
SCOPING REVIEW - Complete This study was led by co-investigator Dr. Florence Cayouette. We reviewed the literature to determine how pediatric assent is defined in research publications, how pediatric assent is operationalized, and variations in assent practices employed.
View the abstract for this study (Coming soon)
STAKEHOLDER SURVEY - Complete We conducted a survey of four key stakeholder groups involved in defining and obtaining assent for research purposes in PICUs in Canada to better understand their perspectives and perceived barriers to assent. The stakeholder groups included in this survey were pediatric critical care nurses, research coordinators and researchers focused on pediatric critical care research, and Canadian REB Chairs/Managers. View the abstract for this study (Coming soon)
PATIENT AND CAREGIVER PERSPECTIVES – Ongoing This multi-centre study is exploring Canadian PICU patient and caregiver perspectives on assent for research in the PICU using survey and interview methodology. Our primary objective is to understand patient and parent/caregiver perspectives to inform operationalization of the assent process for research in the PICU environment. Our secondary objectives are to: (1) obtain patient and parent/caregivers’ thoughts on perceived barriers to the assent process in pediatric intensive care research; and (2) compare the perspectives of patients and parents/caregivers regarding the assent process. We will purposively enroll patients and families with varied demographic characteristics to ensure a diverse and inclusive study cohort. We expect to have results for this study by the end of 2024.
INDIGENOUS YOUTH PERSPECTIVES ON ASSENT IN PICU– Future A future study will specifically focus on Indigenous youth perspectives on assent for research in the PICU, using interview, survey and/or focus group methodology. This study will be conducted in accordance with Chapter 9 of the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (TCPS 2). We will seek community consultation prior to starting study planning and include Indigenous partners and co-investigators throughout the research process.
GUIDELINE DEVELOPMENT AND IMPLEMENTATION STUDY – Future We will conduct a formal international consensus process with stakeholders (including patients and families) from multiple countries to develop international guidelines for assent that are region specific but agree on core principles of the assent process for pediatric critical care research. We will evaluate assent practices across Canada before and after implementation of these guidelines.
Thanks to all Participating Centres and collaborators for engaging in this research program!
Thanks to the CCCTG for endorsing this research project.
Blessing Bazuaye, Naisha Nicolas, Jennifer Armstrong, Lisa Albrecht
Children’s Hospital of Eastern Ontario (CHEO), Alberta Children’s Hospital (Calgary), London Health Sciences Centre (London), Montreal Children’s Hospital (Montreal)
Kusum Menon, Saoirse Cameron, Florence Cayouette, David Garrow, Gary Goldfield, Fatima Mougharbel, Patricia Fontela, Dori-Ann Martin, Sangeeta Mehta, Shauna O’Donnell, Courtney O’Reilly, Angela Wilson
